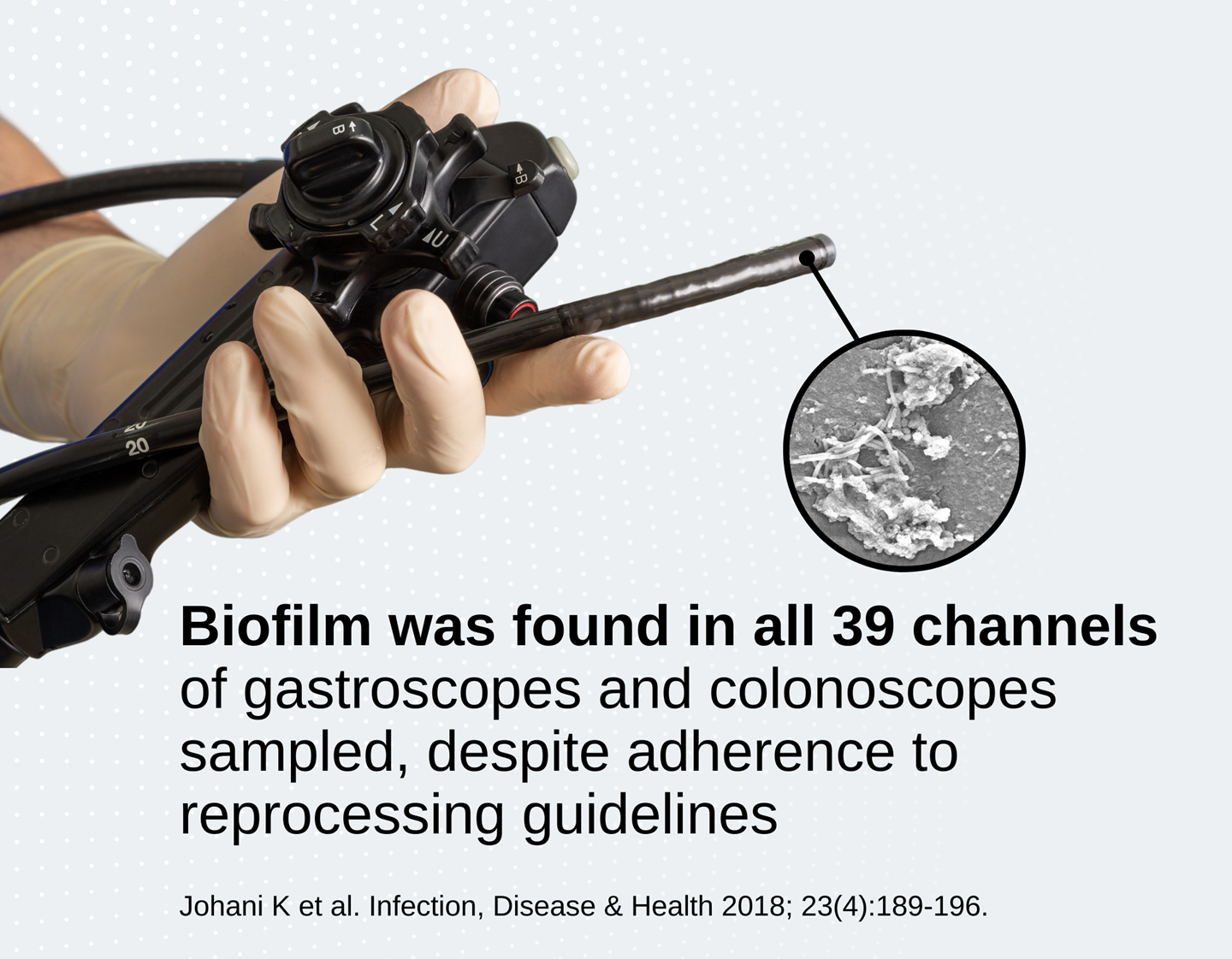
It is essential that biofilm is removed during the manual cleaning stage, as evidence suggests it may be able to survive high-level disinfection.6 This resistance can develop further over time as residual dead cells (biomass) accumulates.7
Resistant strains of biofilm are also emerging. Carbapenem-resistant enterobacterales (CRE) show increased tolerance to peracetic acid (PAA) when grown as a biofilm.8 K. pneumoniae isolated from an outbreak involving a duodenoscope showed weak resistance to PAA in its planktonic (free-living) form. When grown as a biofilm, the bacteria showed resistance to PAA even at a concentration used for high-level disinfection.
Multiple studies have demonstrated that a single strain of bacteria can be transmitted to patients via the same endoscope, over a period of months:
- 27 patients were colonized with multidrug-resistant Klebsiella pneumonia, with 10 developing an active infection. The outbreak was traced to two endoscopes persistently contaminated with K. pneumoniae over 8 months.9
- One endoscope infected multiple patients with the same strain of Pseudonomas aeruginosa. Biofilm was present in undamaged channels and could not be removed despite repeated HLD10
These reports show that endoscopes can act as reservoirs for pathogens, despite repeated cleaning and reprocessing.
Given this evidence, it is likely that the formation of protective biofilms allows pathogens to persist in endoscopes over time.